The antibody-drug conjugate (ADC), a humanized or human monoclonal antibody conjugated with highly cytotoxic small molecules (payloads) through chemical linkers, is a novel therapeutic format and has great potential to make a paradigm shift in cancer chemotherapy. This antibody-based molecular platform enables selective delivery of a potent cytotoxic payload to target cancer cells, resulting in improved efficacy, reduced systemic toxicity, and preferable pharmacokinetics (PK)/pharmacodynamics (PD) and biodistribution compared to traditional chemotherapy.
All three component parts of an ADC, the antibody, the cytotoxic agent, and the linker that joins them, are critical elements in its design. The antibody moiety should be specific for a cell surface target molecule that is selectively expressed on cancer cells, or overexpressed on cancer cells relative to normal cells. The payload of an ADC must be highly cytotoxic so that it can kill tumor cells at the intracellular concentrations achievable following distribution of the ADC into solid tumor tissue, and because only a limited number of payloads can be linked to an antibody molecule (typically an average of 3-4 payloads per antibody) without severely compromising its biophysical and pharmacokinetic properties. The cytotoxic compounds include derivatives of calicheamicin, a class of highly cytotoxic enediyne antibiotics which kill cells by causing DNA double-strand breaks, and derivatives of the potent antimitotic microtubule-disrupting agents, dolastatin 10 (auristatins) and maytansine.
The third vital component of an ADC is the linker that forms a chemical connection between the payload and the antibody. The linker should be sufficiently stable in circulation to allow the payload to remain attached to the antibody while in circulation as it distributes into tissues (including solid tumor tissue), yet should allow efficient release of an active cell-killing agent once the ADC is taken up into the cancer cells. Linkers can be characterized as either cleavable, or as non-cleavable.
| Cat. No. | Product Name | CAS No. | Purity | Chemical Structure |
|---|---|---|---|---|
| KM32393 | AGD-0182 | 1835700-14-5 |
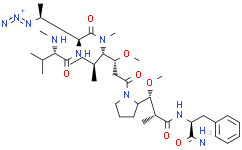
|
|
| KM37423 | Anetumab ravtansine | 1375258-01-7 |

|
|
| KM37423 | Anetumab ravtansine | 1375258-01-7 | 99% |

|
| KM38162 | ARC19499 | 1350667-54-7 |

|
|
| KM38328 | Azintuxizumab vedotin | 1826819-58-2 | 98% |

|
| KM39016 | Belantamab mafodotin | 2050232-20-5 | 98% |

|
| KM32775 | Cantuzumab ravtansine | 868747-45-9 | 98% |

|
| KM36077 | Cofetuzumab pelidotin | 1869937-48-3 | 99% |

|
| KM38156 | Danicamtiv-d3 | 2925925-82-0 |

|
|
| KM38155 | Danicamtiv-d6 | 1970975-54-2 |

|
|
| KM38085 | DBCO-PEG4-VA-PABC-MMAE | 3049211-57-3 | 98% |

|
| KM31829 | Enfortumab vedotin-ejfv | 1346452-25-2 | 98% |

|
| KM36787 | Farletuzumab ecteribulin | 2407465-18-1 | 98% |

|
| KM35454 | Givastomig | 2762499-30-7 | 98% |

|
| KM39026 | Ifinatamab deruxtecan | 2484870-92-8 | 99% |

|
| KM37161 | Ladiratuzumab vedotin | 1629760-29-7 |

|
|
| KM37161 | Ladiratuzumab vedotin | 1629760-29-7 | 99% |

|
| KM38892 | Lifastuzumab vedotin | 1401812-88-1 | 99% |

|
| KM36890 | Lupartumab Amadotin | 1640972-00-4 | 99% |

|
| KM32682 | MASTL-IN-5 | 3024916-78-4 |

|

咨询热线:
400-881-9290